Frequently Asked Questions
Patients within healthcare settings receive medication and other therapies through a variety of tubes and catheters. The connectors of these different delivery systems are often compatible and can be connected to each other. Patient injuries, and even deaths, have occurred when medicines, liquid feeding formulas or air have been accidentally delivered through the wrong tube. These errors are called tubing misconnections.
The ISO 80369 series of Standards aims to avoid misconnection between two unrelated delivery systems (e.g. enteral syringe connected to an IV catheter), which can cause patient injuries or deaths. To do so, ISO 80369 created unique international Standard designs of connectors for each clinical application (Respiratory, Enteral, Urology…), which are non-interconnectable. 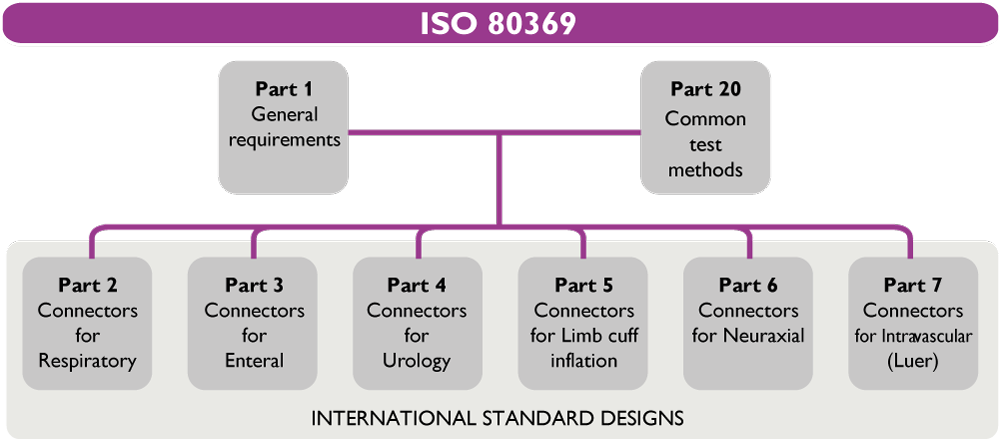
ISO 80369-1 is Part 1 of the ISO 80369 series. Part 1 provides:
- General requirements for small-bore connectors for liquids and gases in healthcare applications
- A framework for testing connectors of different medical applications against each other to ensure non-interconnectability,
- Specific misconnection testing methods
ISO 80369-3 is part 3 of the ISO 80369 series, which is dedicated to enteral applications. Part 3 describes the dimensions, shape, materials and functional requirements of the new proposed enteral standard connector, called ENFit™.
As the inventor of Nutrisafe, one of the first safe enteral feeding systems, Vygon knows how safety in enteral therapy is important and what processes should be implemented to reach the ISO’s objective. That’s why Vygon decided to join the Standard Committee from the start to work on the creation of the ISO 80369 series, particularly for the enteral application.
ENFit™ is the trade name of the new enteral standard connector compliant with ISO 80369-3, in the female to male orientation.
Nutrisafe2 is the name of Vygon’s enteral feeding system dedicated to neonates and newborns, which incorporates the Nutrisafe2 / ISO 80369-1 compliant connectors. This 10-year experienced neonatal system is safe, small and accurate. It minimizes dramatically the risk of volume displacement, down to 0.029 ml*, so the neonatal specialists can use it with confidence.
*GEDSA ENFit® Low Dose Tip Review, Q2 2016
Yes, Nutrisafe2 is compliant with Part 1 of ISO 80369. This means that the design of Nutrisafe2 connectors was assessed to mitigate risks of misconnection to the other connectors from different clinical applications (respiratory, neuraxial, IV….) within ISO 80369.
No, Nutrisafe2 and ENFit™ are not compatible. If you are required to connect these two enteral feeding systems, adaptors would be needed.
Yes, Vygon will maintain all the existing codes for Nutrisafe2.
Vygon continues to promote its Nutrisafe2/ISO 80369-1 compliant enteral feeding system for neonates and newborns, because:
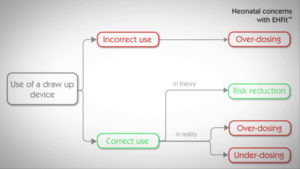
– Using ENFit™ syringes to administrate low dose of enteral medication (up to 2 ml) may result in inaccurate dosing errors (variation of 0.148ml), particularly unacceptable in neonatology.
– Using a Low Dose Tip (LDT) syringe doesn’t eliminate this volume displacement risk. Nutrisafe2, specifically designed for neonatal patients, dramatically reduces this volume displacement risk (division by 4).
As written in the ISO 80369-3:2016, “The volume of the displacement when making a connection matters. In a 500g newborn infant, enteral drugs are often prescribed in volumes as small as 0.1ml or even 0.01ml. “
“Concerns have been raised about the possible risks of delivering accurate doses of medicines in certain clinical practices across high risk subpopulations (e.g. neonatal patients) when using a reversed connection system (female to male). This orientation may introduce inadvertent displacement of fluid originally contained within […] “ the syringe tip.
(Extract from ISO 80369-3:2016)
“Laboratory testing also shows a mid tolerance E1 [ENFit™] connector pair in a female to male orientation displaces a mean average of 0.148ml…” (Extract from ISO 80369-3:2016)
As very low volumes of enteral drugs (0.05ml – 0.1ml) are administered daily to patients in NICU, a high level of accuracy is required. This 0.148ml over-delivery could multiply by a factor of 4 the expected administrated volume and therefore be detrimental for neonatal patients’ health, especially when administrating critical drugs such as morphine, digoxin, methadone, captoril, caffeine, etc.
No, laboratory testing was performed to assess the accuracy of the LDT syringe and the results showed that, in case of using the current practices, the accuracy of the LDT syringe is equivalent to the ENFit™ syringe*, therefore unacceptable for neonates and newborns.
The results appear acceptable only in the case of implementing a new strict syringe protocol**. This new protocol may lead to involuntary dosing errors due to human factors (inadequate training, forgetfulness, skipping a step…) and to microbial & chemical contamination risks.
*Analysis of «the report GEDSA Low Dose Syringe Accuracy test» - SELECT Report - 23 June 2016
**GEDSA ENFit® Low Dose Tip Review, Q2 2016
No:
1. The protocol of using a drawing-up device for sampling is not reliable (see question #20 above).
2. The protocol of removing the fluid from the LDT syringe moat is not accurate and disperses drops of fluid, which introduces the risk of chemical and microbial contamination.
3. Automatic errors due to human factors are unavoidable:
- Linked to routine, distraction, tiredness…
- Occur in common and familiar situations
- Often not immediately noticed
And can lead to dosing inaccuracy.
Moreover, the protocol of removing fluid from the syringe tip can lead to high risk dosing errors (mainly under-dosing) if it is used on another type of syringe (e.g: intravenous Luer syringe)
For Neonatology, Vygon proposes Nutrisafe2, a unique safe enteral feeding system specially designed for the tiniest patients. This 10-year experienced system is safe, small and accurate. It dramatically minimises the risk of volume displacement, down to 0.029 ml*. Neonatal specialists can use it with confidence.
*Theoretical measurements - January 2014 - Internal Data
No, an ISO standard is not a law but an international recommendation. “The important distinction between standards and legislation is that standards are voluntary, whereas legislation is mandatory. When regulatory authorities use standards as a basis for legislation, only then do they become mandatory, and then only within the jurisdiction covered by the legislation. […]” * However, it is strongly recommended to implement the ISO 80369-3 intent in your hospital.
*http://www.iso.org/sites/ConsumersStandards/1_standards.html
The mission of Global Enteral Device Supplier Association (GEDSA) is to help health facilities to understand the new ISO 80369 standard, prepare for this change and implement the non-interconnectable connectors.
Vygon is a charter member of GEDSA.