Dr. O’Mara’s webinar : Dosing precision in the NICU, the Nutrisafe2 syringe
Dr. O’Mara presents the latest results of her work and compares Low Dose Tip syringes and Nutrisafe2 syringes. She highlights the importance of dosing accuracy with enteral syringes used in the NICU. Overdose in this population, which is different from children and adults, is very risky. A simple, reproducible and safe method limits the risk of administration errors.

FDA safety communication
The Food and Drugs Administration (USA) released a safety communication on October 12th, 2021. This safety communication warns patients and healthcare providers about the potential overdoses when using ENFit Low Dose Tip (LDT) syringes due to design of its moat. The communication can be consulted here.
Webinar at jENS 2021
Vygon participated in the joint European Neonatal Societies Virtual Congress in September 2021 where Dr O’Mara presented a symposium. She addressed an important topic in neonatology: are the safety enteral feeding system accurate enough for neonatal drug administration? It is a key question for premature babies in NICUs, Dr O’Mara presented the results of her studies on this particular topic.


The ISO 20695 committee refused to include the LDT syringe in the normative requirements
In September 2017, the first draft of the International Standard was disapproved. One of the main reasons is that the "Low Dose Tip" (LDT) syringe had been integrated into ISO 20695, although its ability to reliably increase dose accuracy is not ensured.
National Coalition for Infant Health (NCfIH) warns about the use of the ENFit system in Neonatology.
As part of the introduction of ENFit on the market, the NCfIH organisation pointed out significant risks associated with the use of the ENFit connector or the Low Dose Tip syringe. In a letter to The Joint Commission, it expressed its concerns about the use of this system on premature newborns, for whom dosage must be of the highest accuracy.
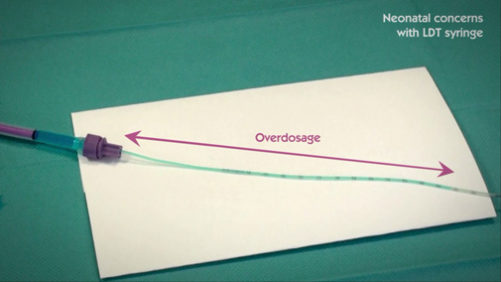
The “Low Dose Tip” syringe can lead to dose inaccuracy
The new syringe design, called Low Dose Tip (LDT) syringe, doesn’t fully address concerns about dose accuracy in neonates. Laboratory testing showed inconclusive results.