Scientific evidence on accuracy
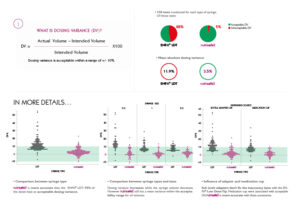
The accuracy of ENFit LDT syringes and nutrisafe2 syringes was compared using Dosing Variance calculation. (Comparison of Dosing Accuracy Between the ENFit LDT and a Neonatal-Specific ISO-Compliant Enteral Syringe – Dr Keliana O’Mara et al)
FDA safety communication
The ENFit Low Dose Tip was created to solve the accuracy concern raised by the standard ENFit connection.
The Food and Drugs Administration (USA) released a safety communication on October 12th, 2021. This safety communication warns patients and healthcare providers about the potential overdoses when using ENFit Low Dose Tip (LDT) syringes due to design of its moat. The communication can be consulted here.
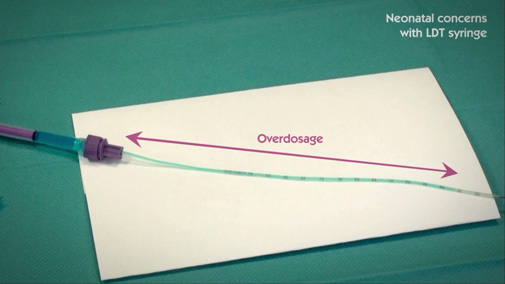
Low Dose Tip (LDT) syringe doesn’t ensure dose accuracy
Laboratory testing has shown that the Low Dose Tip syringe moat could be unintentionally filled with fluid during sampling.
Download
- Comparison of Dosing Accuracy Between the ENFit LDT and a Neonatal-Specific ISO-Compliant Enteral Syringe
- Report on the overdosing risk : ENFit / Low Dose Tip syringe / Nutrisafe2
Focus on the Low Dose Tip (LDT) syringe instructions for use
Click here to see the LDT syringe instructions.
Human errors inevitably happen
Human errors are inadvertent and unintentional, and they happen predictably with repeated acts because the person is on “automatic mode”. Unfortunately, medication errors also increase when there is a higher severity of illness and a higher level of care being given.
The addition of new and specific steps in the syringe workflow process, only during the use of the LDT syringe, will inevitably lead to errors due to:
- distractions
- increase in workload
- “automatic mode” with common and familiar tasks
It’s unfortunate that, when “automatic errors” occur, they are often not immediately noticed due to the nature of the care giver being on “autopilot”. Education is successful in mitigating the risks and learning new protocols but after the training event the individual’s knowledge retention drops on the “forgetting curve“. In order to ensure continued compliance, this training must be continuous and sustainable especially as care givers are working in stressful situations.
Concerns that the LDT instructions could lead to overdose
- The use of a drawing-up device not only increases the hospital budget, it also doesn’t guarantee that the syringe moat is clear of fluid (See the use of a draw-up device video).
- Tapping or flicking the syringe to remove the excess fluid from the syringe moat results in droplets being dispersed and introduces the risk of chemical and microbial contamination of the operator and the dispensing area.
- Removing the fluid from the LDT syringe moat can be difficult, depending on the fluid viscosity.
- The wiping method to clear the syringe moat can be difficult to perform as the moat of the LDT syringe is thin and deep. Moreover, the towel used to wipe the syringe moat must ensure no contamination risk is introduced of particles or microorganisms
- If the LDT syringe instructions for use are used with another type of syringes (e.g. classic ENFit, intravenous Luer syringe, neuraxial syringe…), it would lead to underdosing errors for the clinical application.