What is dose inaccuracy?
To understand the challenges of NICUs regarding drug administration with ENFitTM you need to know what dose inaccuracy is and where it comes from.
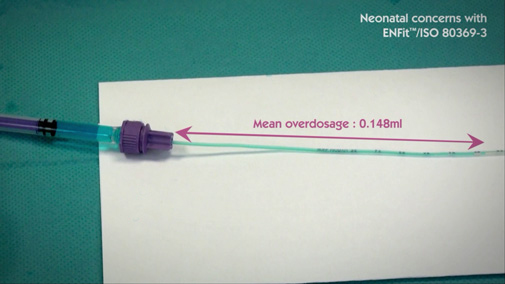
ENFit™ can lead to inaccurate dosage
As written in the ISO 80369-3, “Laboratory testing shows a mid-tolerance [ENFit™] connector pair in a female to male orientation displaces a mean average of 0.148ml (min 0.089ml and max 0.179ml with an n = 32) of fluid. “
This volume displacement is particularly unacceptable for oral medications administrated in low doses to neonatal patients.

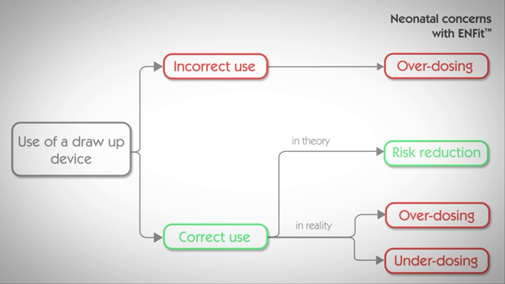
A drawing-up device can’t eliminate the ENFit™ overdosing risk
If we assume the drawing-up protocol is correctly followed, the tension surface effect and/or the viscosity of the medication can still lead to a variation in the dose delivered (up to 0.148ml).
The volume displacement risk can’t be eliminated